1Department of Smart Agriculture Systems Machinery Engineering, Chungnam National University, Daejeon, Republic of Korea
2Department of Agriculture Engineering, National Institute of Agricultural Science, Rural Development Administration, Wanju, Republic of Korea
3Department of Smart Agriculture Systems, College of Agricultural and Life Science, Chungnam National University, Daejeon, Republic of Korea
Correspondence to Byoung-Kwan Cho, E-mail: chobk@cnu.ac.kr
Plant Image Sci. 1:2. https://doi.org/10.65971/PIS.2025.1.2
Received on November 09, 2025, Revised on December 30, 2025, Accepted on December 30, 2025, Published on December 31, 2025.
© Author(s). This is an Open Access article distributed under the terms of the Creative Commons CC BY 4.0 (https://creativecommons.org/licenses/by/4.0/) which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Imaging technologies have become indispensable tools in modern plant phenotyping, transforming visual information into measurable traits essential for analyzing morphology, physiology, biochemistry, and micro- to nanoscale structures. This concise review summarizes recent advances by dividing plant imaging into two major categories: (1) physiological and biochemical, which includes hyperspectral, multispectral, and fluorescence hyperspectral imaging, as well as terahertz imaging, surface-enhanced Raman scattering, and carbon dot-based techniques; and (2) structural and morphological, encompassing RGB, thermal, light detection and ranging (LiDAR), confocal microscopy, and optical coherence tomography. Together, these modalities deliver insights from the canopy to the molecular level, enabling precise monitoring of plant stress, disease, and developmental traits. By integrating these multimodal imaging techniques with artificial intelligence, the review highlights key developments, current challenges, and future perspectives in plant measurement and analysis.
artificial intelligence, molecular-scale imaging, phenotyping, spectral imaging, structural imaging
The combined impacts of population growth and climate change necessitate a 100–110% increase in global crop yields by 2050 (Tilman et al. 2011), underscoring the urgent need to improve agricultural efficiency and sustainability. To meet this challenge amid climate stresses, such as heat and drought, agricultural research is shifting from traditional, destructive methods to advanced plant imaging technologies that are indispensable tools for modern phenotyping. These technologies quantify essential plant traits across morphology, physiology, and biochemistry, ranging from the macro-to the nanoscale. This review categorizes recent imaging progress into two analytical domains: (1) physiological and biochemical trait assessment, which includes multispectral, fluorescence, hyperspectral, and nanomaterial-based approaches (terahertz, SERS, and Carbon dots); and (2) structural and morphological monitoring, which includes RGB, thermal imaging, unmanned aerial vehicle (UAV) drones, light detection and ranging (LiDAR), confocal microscopy, and optical coherence tomography. In addition to these domains, this review addresses the integration of artificial intelligence with multimodal s trategies, challenges in data standardization, and future directions for unified data-driven phenotyping platforms.
These approaches enable non-destructive monitoring of key metabolites and their responses to environmental factors, including drought, heat, nutrient status, and photosynthetic dynamics. These techniques are essential for investigating plant functions at the macro-, micro-, and nanoscale levels. This section is divided into two parts: (i) Spectral imaging and (ii) molecular-scale biosensing. These techniques are essential for investigating plant functions at the macro-, micro-, and nanoscale levels.
Spectroscopic technologies analyze how light interacts with plant tissues through reflectance, absorbance, and transmittance, revealing key biochemical components, such as chlorophyll, phenylpropanoids, proteins, and water. Infrared (IR), near-infrared (NIR), and Raman spectroscopies detect functional groups (e.g., N–H, O–H, and C–H), offering detailed chemical insights, but limited spatial resolution. To address this issue, spectral imaging technologies combine spectral and spatial information for a comprehensive plant analysis. Hyperspectral imaging (HSI) captures hundreds of contiguous bands to form a hypercube, enabling precise profiling of nutrients and stress-related metabolites such as phenolics and flavonoids (Jayapal et al. 2022). Multispectral imaging (MSI) records fewer bands (3-15) and is widely used to compute vegetation indices such as normalized difference vegetation index (NDVI) to assess plant health (Vuletić et al. 2023). Fluorescence hyperspectral imaging integrates fluorescence and hyperspectral data to monitor changes in chlorophyll fluorescence, thereby providing insights into photosynthetic efficiency (Faqeerzada et al. 2022). Collectively, these high-throughput technologies deliver multidimensional data that are essential for assessing plant physiological responses to drought and heat stress. By monitoring variations in key metabolites such as chlorophyll and phenolics, as well as imaging-derived indicators related to water-use efficiency, these approaches provide valuable insights into plant resilience under variable climate conditions (Beegum et al. 2024; Roy et al. 2023).
Spectral imaging provides valuable spatial and spectral insights; however, its sensitivity and spatial resolution can fall short of micro- and nanoscale measurements. These constraints can be overcome using molecular-scale biosensing technologies that enable highly sensitive molecular characterization. Surface-enhanced Raman scattering (SERS) amplifies Raman signals using gold or silver nanostructures to precisely map pesticides, pathogens, and metabolites (Fleischmann et al. 1974; Huang et al. 2025; Ma et al. 2015). Terahertz (THz) imaging exploits the radiation between infrared and microwave frequencies, providing a noninvasive assessment of plant water status and crop yield estimation (Federici et al. 2009; Zang et al. 2019). Carbon dots (CDs) are nanoscale fluorescent materials that support nutrient tracking and chemical detection with high biocompatibility (Bhattacharya et al. 2024; Liu et al. 2020; Maholiya et al. 2023). Together, these methods provide high-resolution molecular-level insights that are crucial for precision agriculture.
Biochemical imaging provides valuable physiological information but offers limited insight into structural traits such as plant height, crown diameter, and architecture. To address this gap, structural and morphological imaging methods are essential and broadly divided into two main categories: (i) two-dimensional (2D) imaging technologies and (ii) three-dimensional (3D) imaging technologies, from the macroscale to the microscale.
Two-dimensional imaging has long been central to plant phenotyping because of its simplicity, accessibility, and cost-effectiveness. Among these, RGB imaging is the most widely used and employs digital cameras to capture red, green, and blue light, enabling detection of phenotypic differences between healthy and stressed plants (Pérez-Bueno et al. 2016). This high-resolution method enables real-time monitoring of growth and developmental traits; however, its accuracy declines in dense canopies due to leaf overlap and occlusion, thereby hindering organ-level segmentation. Thermal imaging complements RGB by measuring the canopy surface temperature, offering insights into plant water status, heat stress, and disease symptoms. Stressed plants often display distinct thermal signatures compared to healthy controls, although thermal imaging is limited by its low spatial resolution and sensitivity to ambient environmental conditions (Pérez-Bueno et al. 2016). UAV platforms have further enhanced fieldscale phenotyping by integrating RGB and thermal sensors for the large-area monitoring of canopy temperature, chlorosis, pest distribution, and irrigation efficiency (Toscano et al. 2024). Collectively, these 2D imaging technologies underpin the development of scalable high-throughput phenotyping systems.
Although 2D imaging techniques offer rapid and efficient measurements, they are limited in their ability to capture complex plant architectures due to self-occlusion and overlapping leaves. 3D imaging addresses these challenges by enabling detailed analysis of plant morphology, including volume, density, leaf arrangement, and stem organization. LiDAR is a prominent 3D technique that uses pulsed lasers to generate dense point clouds and provides precise measurements of plant height, canopy volume, leaf area, and stem spacing. Its effectiveness has been demonstrated by large-scale phenotyping in sorghum (Patel et al. 2023). For tissue-level characterization, confocal microscopy offers high-resolution 3D visualization of cells and tissues, revealing plant-microbe interactions and developmental processes (Nelson et al. 2024; Harline and Roeder 2023). Optical coherence tomography (OCT) provides nondestructive cross-sectional imaging at micrometer resolution, enabling internal tissue observation and disease monitoring (Fujimoto et al. 2000; Sasi and Chauvet 2025). Collectively, these macroscale and microscale 3D methods provide hierarchical morphological insights that are vital for comprehensive plant phenotyping.
AI has revolutionized plant imaging by processing massive datasets across diverse modalities, including RGB, thermal, hyperspectral, multispectral, fluorescence, 3D imaging, SERS, THz, and CDs. AI enhances data interpretation, robust feature extraction, and precise classification of plant health indicators, including stress, disease, and biochemical traits. Machine learning excels at estimating chlorophyll levels, detecting pesticide residues, and tracking biochemical changes (Bhattacharya et al. 2024; Faqeerzada et al. 2022; Jayapal et al. 2022). Deep learning analyzes complex 3D and high-dimensional data for accurate segmentation and morphological insights (Jin et al. 2025; Patel et al. 2023). Multimodal data integration via AI enables scalable high-throughput phenotyping systems that are essential for precision agriculture (Yang et al. 2025). AI-driven fusion improves trait analysis, minimizes human error, and supports reliable decision making. Fig. 1 shows a schematic representation of these advanced plant-imaging techniques across all four domains.
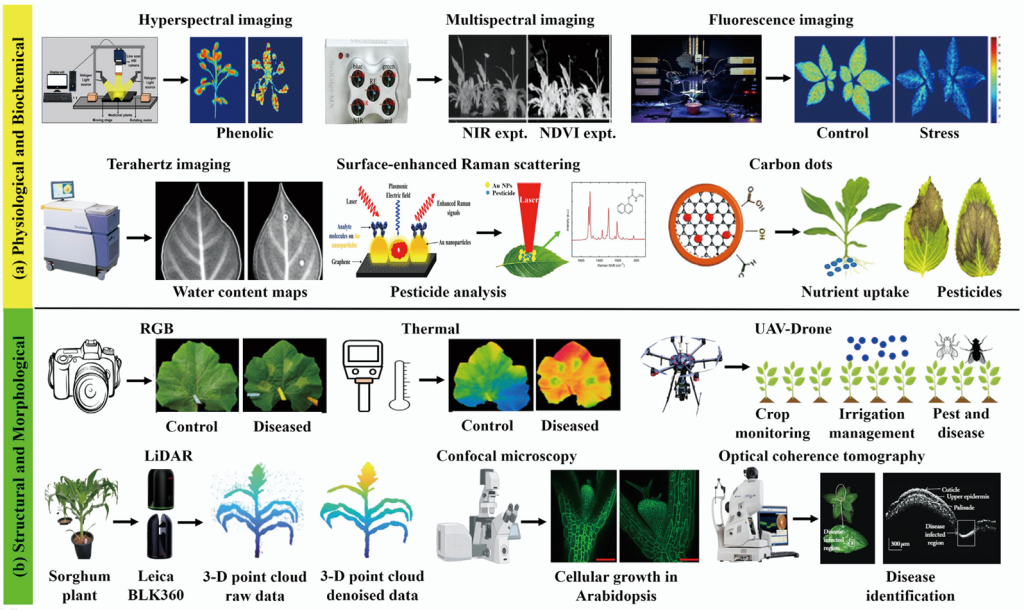
Fig. 1. Imaging and sensing modalities for plant phenotyping using (a) physiological/biochemical methods and (b) structural/ morphological methods. Images adapted from literature cited in the text. NIR, near-infrared; NDVI, normalized difference vegetation index; SERS, surface-enhanced Raman scattering; RGB, red-green-blue; UAV, unmanned aerial vehicle; LiDAR, light detection and ranging; 3-D, three-dimensional; OCT, optical coherence tomography.
The imaging techniques discussed, spectral, structural, molecular, and AI-driven, collectively enable comprehensive plant phenotyping, capturing biochemical traits, morphological features, cellular-level details, and effective multimodal data fusion. Despite these strengths, key challenges remain in achieving real-time measurements, data standardization, and scalable field deployment. Future investigations should aim to unify the complementary modalities within AI-enhanced multiscale platforms. Continued progress in standardization, hardware optimization, and multimodal data fusion is essential to enable real-time and resilient phenotyping systems in precision agriculture.
This review provides an overview of current plant imaging approaches and grouping technologies into physiological, biochemical, and structural/morphological domains. Although spectral imaging (HSI, MSI, and fluorescence) captures vital chemical signatures, it is limited by high costs and the need for intensive data processing. Biosensing modalities (SERS, THz, and CDs) offer nanoscale sensitivity for stress detection, but struggle with reproducibility. Furthermore, although 2D systems are prone to environmental interference and occlusion, 3D techniques (LiDAR, confocal, and OCT) provide superior structural resolution at the expense of portability and acquisition speed. Together, these tools form a robust phenotyping foundation, providing the data-driven insights necessary for precision agriculture.
Methodology: Joshi R, Faqeerzada MA, Kim T; Investigation: Joshi R, Faqeerzada MA, Kim T; Supervision: Cho BK; Writing – original draft: Joshi R, Faqeerzada MA, Cho BK; Writing – review and editing: Joshi R, Faqeerzada MA, Kim T, Cho BK.
The authors have no conflict of interest.
Not applicable.
This work was supported by the Korea Institute of Planning and Evaluation for Technology in Food, Agriculture, and Forestry (IPET) through the Smart Farm Innovation Technology Development Program, funded by the Ministry of Agriculture, Food, and Rural Affairs (MAFRA)(421030-04).
Not applicable.
Beegum S et al. 2024. Hyperspectral reflectance-based high throughput phenotyping to assess water-use efficiency in cotton. Agriculture. 14(7):1054. https://doi.org/10.3390/agriculture14071054
Bhattacharya T et al. 2024. l-Cysteine-modified carbon dots derived from Hibiscus rosa-sinensis for thiram pesticides identification on edible perilla leaves. ACS Omega. 9(48):47647–47660. https://doi.org/10.1021/acsomega.4c07090
Faqeerzada MA et al. 2022. Fluorescence hyperspectral imaging for early diagnosis of heat-stressed ginseng plants. Appl Sci. 13(1):31. https://doi.org/10.3390/app13010031
Federici JF, Wample RL, Rodriguez D, Mukherjee S. 2009. Application of terahertz Gouy phase shift from curved surfaces for estimation of crop yield. Appl Opt. 48(7):1382–1388. https://doi.org/10.1364/ao.48.001382
Fleischmann M, Hendra PJ, McQuillan AJ. 1974. Raman spectra of pyridine adsorbed at a silver electrode. Chem Phys Lett. 26(2):163–166. https://doi.org/10.1016/0009-2614(74)85388-1
Fujimoto JG, Pitris C, Boppart SA, Brezinski ME. 2000. Optical coherence tomography: an emerging technology for biomedical imaging and optical biopsy. Neoplasia. 2(1–2):9–25. https://doi.org/10.1038/sj.neo.7900071
Harline K, Roeder AHK. 2023. An optimized pipeline for live imaging whole Arabidopsis leaves at cellular resolution. Plant Methods. 19:10. https://doi.org/10.1186/s13007-023-00987-2
Huang Q, Guo H, Wang W, Kang S, Vikesland PJ. 2025. Surface-enhanced Raman spectroscopy (SERS) based biological and environmental 2D and 3D imaging. ACS Environ Au. 5(4):342–362. https://doi.org/10.1021/acsenvironau.4c00149
Jayapal PK et al. 2022. Non-destructive measurement of total phenolic compounds in Arabidopsis under various stress conditions. Front Plant Sci. 13:982247. https://doi.org/10.3389/fpls.2022.982247
Jin S et al. 2025. Deep learning for three-dimensional (3D) plant phenomics. Plant Phenomics. 7(4):100107. https://doi.org/10.1016/j.plaphe.2025.100107
Liu J, Li R, Yang B. 2020. Carbon dots: a new type of carbon-based nanomaterial with wide applications. ACS Cent Sci. 6(12):21792195. https://doi.org/10.1021/acscentsci.0c01306
Ma B, Li P, Yang L, Liu J. 2015. Based on time and spatial-resolved SERS mapping strategies for detection of pesticides. Talanta. 141:1–7. https://doi.org/10.1016/j.talanta.2015.03.053
Maholiya A et al. 2023. An insight into the role of carbon dots in the agriculture system: a review. Environ Sci Nano. 10(4):959–995. https://doi.org/10.1039/D2EN00954D
Nelson AC et al. 2024. Assembly and evaluation of a confocal microscopy image analysis pipeline useful in revealing the secrets of plant-fungal interactions. Mol Plant Microbe Interact. 37(12):804–813. https://doi.org/10.1094/MPMI-08-24-0090-TA
Patel AK et al. 2023. Deep learning-based plant organ segmentation and phenotyping of sorghum plants using LiDAR point cloud. IEEE J Sel Top Appl Earth Obs Remote Sens. 16:8492–8507. https://doi.org/10.1109/JSTARS.2023.3312815
Pérez-Bueno ML, Pineda M, Cabeza FM, Barón M. 2016. Multicolor fluorescence imaging as a candidate for disease detection in plant phenotyping. Front Plant Sci. 7:1790. https://doi.org/10.3389/fpls.2016.01790
Roy B et al. 2023. Early detection of drought stress in durum wheat using hyperspectral imaging and photosystem sensing. Remote Sens. 16(1):155. https://doi.org/10.3390/rs16010155
Sasi GS, Chauvet AAP. 2025. Using optical coherence tomography in plant biology research: review and prospects. Sensors. 25(8):2467. https://doi.org/10.3390/s25082467
Tilman D, Balzer C, Hill J, Befort BL. 2011. Global food demand and the sustainable intensification of agriculture. Proc Natl Acad Sci. 108(50):20260–20264. https://doi.org/10.1073/pnas.1116437108
Toscano F et al. 2024. Unmanned aerial vehicle for precision agriculture: a review. IEEE Access. 12:69188–69205. https://doi.org/10.1109/ACCESS.2024.3401018
Vuletić J, Car M, Orsag M. 2023. Close-range multispectral imaging with Multispectral-Depth (MS-D) system. Biosyst Eng. 231:178194. https://doi.org/10.1016/j.biosystemseng.2023.06.002
Yang ZX, Li Y, Wang RF, Hu P, Su WH. 2025. Deep Learning in multimodal fusion for sustainable plant care: a comprehensive review. Sustainability. 17(12):5255. https://doi.org/10.3390/su17125255
Zang Z, Wang J, Cui HL, Yan S. 2019. Terahertz spectral imaging based quantitative determination of spatial distribution of plant leaf constituents. Plant Methods. 15:106. https://doi.org/10.1186/s13007-019-0492-y